Media Release
Deakin materials scientists have proven that liquid solvent-free solid state batteries can easily be made, paving the way for a future free from batteries catching fire or exploding if they get too hot.
Research fellows at Deakin’s Institute for Frontier Materials Dr Fangfang Chen and Dr Xiaoen Wang say their discovery means that lithium–ion batteries found in everyday items like mobile phones, computers and vehicles would no longer pose a fire risk because the highly volatile liquid electrolyte currently used in them would be replaced with a solid polymer material.
“This investigation has the potential to underpin some very significant work to improve batteries at two key points,” Dr Wang said.
“Our findings suggest that next generation batteries will be much safer and have exceptionally better performance. From what we’ve discovered, this electrolyte will allow us to use a lithium metal anode, which could see future batteries last twice as long as they currently do with one charge. Alternatively, batteries could end up half of their size and weight without compromising performance time.”
Dr Chen believes that the findings could change the way batteries are handled in everyday life too.
“If industry implements our findings I see a future where battery reliant devices can be safely packed in airplane baggage, for example, or where electric cars don’t pose a fire risk for occupants or emergency services like they currently do,” Dr Chen said.
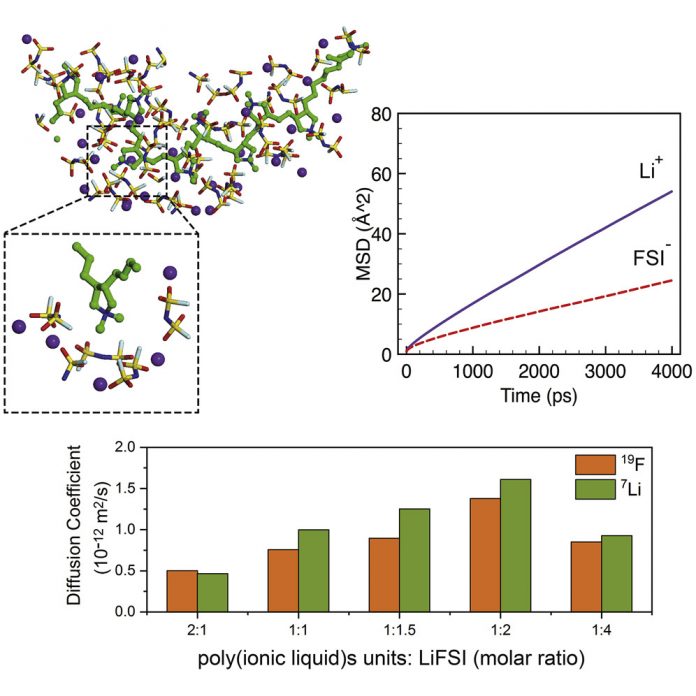
Current lithium-ion batteries are highly flammable and the predominant cause of fires in battery reliant products.
“We’ve reinvented the way polymer interacts with lithium salt, removing the normally highly flammable properties of traditional lithium batteries,” Dr Chen said.
The researchers say they’ve used existing commercial polymer materials to create the new process, meaning the formulation could be applied commercially with little difficulty.
“All of the products that we’ve used to make this safer battery process already exist in the market. Polymers have been used as battery conductors for over 50 years, but we’re the first to use existing commercial polymer in an improved way,” Dr Wang said.
“We’ve done this by weakly bonding the lithium ion with polymer, creating solid polymer electrolytes. We believe this is the first clear and useful example of liquid-free and efficient transportation of lithium-ion in the scientific community.”
So far they’ve proven the process in coin cell batteries, similar to a watch battery size, with the next step being to scale up the batteries to bigger applications – such as for a mobile phone.
“We have started the pouch cell fabrication and testing at Deakin’s word-class Battery Technology Research and Innovation Hub at Waurn Ponds. Once we achieve pouch size, we hope to attract collaboration with industry partners,” Dr Wang said.
‘Poly(Ionic Liquid)s-in-Salt-Electrolytes with Coordination-Assisted Lithium-Ion Transport for Safe Batteries’ was recently published in the journal Joule.




















